


xxxxxThe English scientist John
Dalton put forward his chemical atomic theory in his New
System of Chemical Philosophy, published in 1808. Based
on his research into the nature of gases, he claimed that the atoms
which made up matter were of differing weights and combined together
in definite proportions. Thus chemical reaction was the
rearrangement of these tiny particles from one form of combination
to another. In support of this theory, he produced a table showing
the atomic weights of all the elements known at that time, using a
scale based on the value of hydrogen. Some of his findings were
inaccurate, but the procedure he adopted made it possible to arrange
elements in accordance with their atomic weights -
JOHN DALTON 1766 -
(G3a, G3b, G3c, G4, W4, Va)
Acknowledgements
Dalton: by the
English engraver Charles Turner (1774-
 xxxxxThe English physicist and chemist John
Dalton was self-
xxxxxThe English physicist and chemist John
Dalton was self-
 xxxxxIn 1808 Dalton published his
atomic theory in his New System of Chemical
Philosophy, and listed the first tentative calculations of
the atomic weights of all the elements known at that time. As all
these weights were relative, and a standard was therefore required,
he used a scale based on the atomic weight of hydrogen as one. Some
of his findings were by no means accurate, but the procedure was
sound, and proved the first attempt at compiling today’s periodic
table of the elements. It was now possible to establish order by
arranging elements in accordance with their atomic weights and their
reactions. This was a major achievement.
xxxxxIn 1808 Dalton published his
atomic theory in his New System of Chemical
Philosophy, and listed the first tentative calculations of
the atomic weights of all the elements known at that time. As all
these weights were relative, and a standard was therefore required,
he used a scale based on the atomic weight of hydrogen as one. Some
of his findings were by no means accurate, but the procedure was
sound, and proved the first attempt at compiling today’s periodic
table of the elements. It was now possible to establish order by
arranging elements in accordance with their atomic weights and their
reactions. This was a major achievement.
xxxxxDalton’s investigation
into the physical properties of air and other gases -

xxxxxLikewise, his long and close study of weather conditions also resulted in a number of advances in meteorology, contained in his Meteorological Observations and Essays of 1793. Following a study of evaporation and humidity, for example, he produced a table of vapour pressures of water at various temperatures, and he was the first to substantiate that rain was caused by a fall in temperature, and was not due to a change in atmospheric pressure, as many had believed. And after close observation over a long period, he came to the conclusion that the aurora borealis must have some connection with the Earth’s magnetism.
xxxxxDalton was born in Eaglesfield, near Cockermouth in Cumberland, the son of a weaver. He attended a Quaker school in the town, and began teaching there at the age of 12. After ten years or more in Kendal, running a school with his elder brother, he settled in Manchester in 1793 and remained there, working as a teacher and lecturer in mathematics and chemistry. Though he had no formal training in chemical research, he carried out his investigations with immense dedication and application, and, being professionally independent, seldom accepted data that he himself had not “proven”. He was a quiet, unassuming man, and he made few friends. Such was the esteem in which he was held, however, that he was invited to run courses at the Royal Institution in London in the early 1800s, was made a fellow of the Royal Society in 1822, and awarded the society’s gold medal four years later. In 1830 he became an associate member of the French Academy of Sciences. Today he is generally regarded as the founder of modern atomic theory.

xxxxxIncidentally, the idea of matter being made up of atoms goes back well over 2,000 years. The Greek philosophers Leucippus and Democritus of the 5th century BC, and Epicurus of the 3rd century BC all came up with the idea of “atomism”. Democritus (illustrated), who taught at Abdera in Thrace, held that matter was not continuous but made up of invisible “pieces”. Only atoms and empty space existed, everything else was “superficial appearance”. ……
xxxxx…… Dalton was one of the first scientists, if not the first, to give a description of colour blindness. This was contained in a paper he delivered to the Manchester Literary and Philosophical Society, and published in 1794 under the title Extraordinary Facts Relating to the Vision of Colours. He himself suffered from this impairment of vision (as did his brother). His explanation as to the cause of this condition was way off the mark, but nevertheless colour blindness is often known as “Daltonism”.
Including:
Jöns Berzelius and
Thomas Young

 xxxxxThe Swedish chemist Jöns
Berzelius (1779-
xxxxxThe Swedish chemist Jöns
Berzelius (1779-
 xxxxxBerzelius was born near Linkoping in Ostergotland,
and studied science and medicine at Uppsala University. In 1807 he
was appointed professor of medicine, botany and pharmacy at the
College of Medicine, Stockholm, and from 1815 to 1832 was
professor of chemistry at the Royal Caroline Medico-
xxxxxBerzelius was born near Linkoping in Ostergotland,
and studied science and medicine at Uppsala University. In 1807 he
was appointed professor of medicine, botany and pharmacy at the
College of Medicine, Stockholm, and from 1815 to 1832 was
professor of chemistry at the Royal Caroline Medico-
xxxxxDuring his
career, he isolated and identified a number of new elements,
including selenium in 1817, silicon in 1823, and thorium five
years later. Otherxchemists at this time
who contributed in this way were the German Martin
Klaproth (1743-
xxxxxIncidentally, the terms “protein” and “catalyst” were coined by
Berzelius, and he was one of the first scientists of this period
to group together elements of similar chemical properties. For
example, he introduced the collective term “halogens” (salt
formers) to describe chlorine, bromine and iodine -

xxxxx…… ThexWollaston Medal, the highest award of the Geological Society of London, was funded with a bequest by the English chemist William Hyde Wollaston, and was first awarded in 1831. It was originally made of palladium, one of the metals he discovered, but it is now made in gold. The first recipient was the English geologist William Smith.
xxxxxThe Swedish chemist Jöns Berzelius (1779-
G3c-
xxxxxLike Dalton, the English
physicist and physician Thomas Young (1773-
 xxxxxLike Dalton, the English physicist and physician Thomas Young (1773-
xxxxxLike Dalton, the English physicist and physician Thomas Young (1773- astigmatism (short-
astigmatism (short-
xxxxxIn 1802 Young also began a study of light, and by demonstrating the phenomenon of optical interference established the wave nature of light. Later, in 1817, this led to his explanation of polarisation, based on the proposition that light waves vibrate at right angles to the direction of travel (i.e. they are “transverse”), and not along the line of travel (“longitudinal), as previously assumed. Among other research projects, he spent time on measuring the size of molecules and on studying elasticity, a branch of physics in which an absolute measurement is named “Young’s modulus” in his honour.
xxxxxHe was born in Milverton,
Somerset, and studied at the universities of London, Edinburgh, and
Göttingen. Apart from producing works on physics and medicine, he
also wrote on Egyptology, and, beginning in 1814, made some headway
in deciphering the hieroglyphics on the Rosetta Stone. It was the
French linguist and historian Jean François
Champollion, however, who
eventually succeeded in producing a full translatio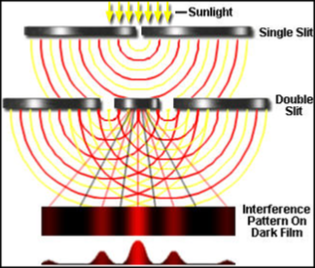 n
in 1822 (G4).
n
in 1822 (G4).
xxxxxIncidentally,
Young’s wave theory of light, valid though it was, was not
convincingly explained or proven, and, being in opposition to
Newton’s theory, failed to gain general acceptance. It was left to
the French physicists Augustin-


